The Science Behind Nuclear Bombs
The Atom
Fun Fact: Did you know that you are not touching anything at any given time? Because due to the negative properties of electrons, atoms repel each other like magnets.
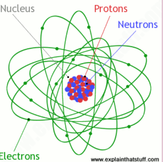
...by definition is the most basic form of matter and the definition of matter is anything that takes up space and has mass. The parts of an atom include protons (positive charge), neutrons (neutral charge), and electrons (negative charge. The protons and neutrons are in center of the atom and make up the nucleus, but the electrons move around the outside of the atom in an electron cloud.
Fun Fact: Did you know that you are not touching anything at any given time? Because due to the negative properties of electrons, atoms repel each other like magnets.
...by definition is the most basic form of matter and the definition of matter is anything that takes up space and has mass. The parts of an atom include protons (positive charge), neutrons (neutral charge), and electrons (negative charge. The protons and neutrons are in center of the atom and make up the nucleus, but the electrons move around the outside of the atom in an electron cloud.
Isotopes and Radioactive Decay
Fun Fact: Did you know that when isotopes return to their stable state, it's called radioactive decay? In relation to carbon, the stage of the atom in radioactive decay is how they do carbon dating.
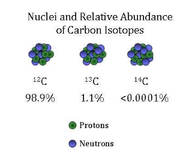
Isotopes are atoms that either have an extra neutron or or a missing neutron. For example, carbon-12 has 6 neutrons and 6 electrons (the most common), but carbon-14 has 8 neutrons and 6 electrons.
Fun Fact: Did you know that when isotopes return to their stable state, it's called radioactive decay? In relation to carbon, the stage of the atom in radioactive decay is how they do carbon dating.
Isotopes are atoms that either have an extra neutron or or a missing neutron. For example, carbon-12 has 6 neutrons and 6 electrons (the most common), but carbon-14 has 8 neutrons and 6 electrons.
Fission and Fusion
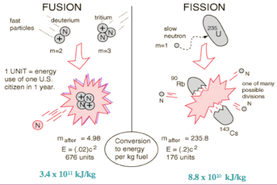
Fission is when you split something into two or more parts, and in science it’s when you split an atom with a neutron. Fusion is when two or more things is combined into one object, and with nuclear fusion it’s the act of combining two different atoms together in order to form another atom.
Fission is when you split something into two or more parts, and in science it’s when you split an atom with a neutron. Fusion is when two or more things is combined into one object, and with nuclear fusion it’s the act of combining two different atoms together in order to form another atom.
The Types of Bombs Dropped on Hiroshima and Nagasaki
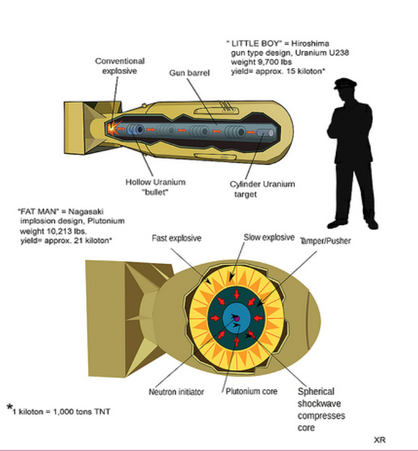
The names of the bombs dropped on the two Japanese cities are called “Fat Man” and “Little Boy.” Fat Man was a “implosion bomb” type and used the isotope plutonium-240. The outermost area of the bomb’s core was made of strong explosives, an absorber shell, the uranium reflector shell, the plutonium pit , and the neutron initiator. The explosives would go off and press the central spheres together and charge the uranium. The pressure created by the explosions made the plutonium reach its critical mass, making it fire off neutrons. The isotope uranium-235 used was then hit by the neutron and split into two atoms. The splitting of the atom makes two atoms, barium and krypton. The fission then creates high amounts of energy, creating the explosion. The reaction continues until there is no more uranium, because the fission process creates two more neutrons which continues to split uranium atoms.
The names of the bombs dropped on the two Japanese cities are called “Fat Man” and “Little Boy.” Fat Man was a “implosion bomb” type and used the isotope plutonium-240. The outermost area of the bomb’s core was made of strong explosives, an absorber shell, the uranium reflector shell, the plutonium pit , and the neutron initiator. The explosives would go off and press the central spheres together and charge the uranium. The pressure created by the explosions made the plutonium reach its critical mass, making it fire off neutrons. The isotope uranium-235 used was then hit by the neutron and split into two atoms. The splitting of the atom makes two atoms, barium and krypton. The fission then creates high amounts of energy, creating the explosion. The reaction continues until there is no more uranium, because the fission process creates two more neutrons which continues to split uranium atoms.
Now for the Little Boy bomb, uranium-235, an isotope of uranium-238 was used. The mechanism involved the following, explosives at the beginning of the tube, a hollow uranium bullet, a barrel, a neutron initiator and then a mass of uranium at the end of the the barrel. The explosives would fire, sending the bullet off at high speeds towards the uranium mass at the end, making them reach critical mass, the neutron initiator then fired off a neutron and made the fission process begin. Krypton and barium was made from the splitting of the uranium atoms, and the energy from the atom splitting, created another explosion.
